You must be logged in to post a review.
SYNVISC ONE®
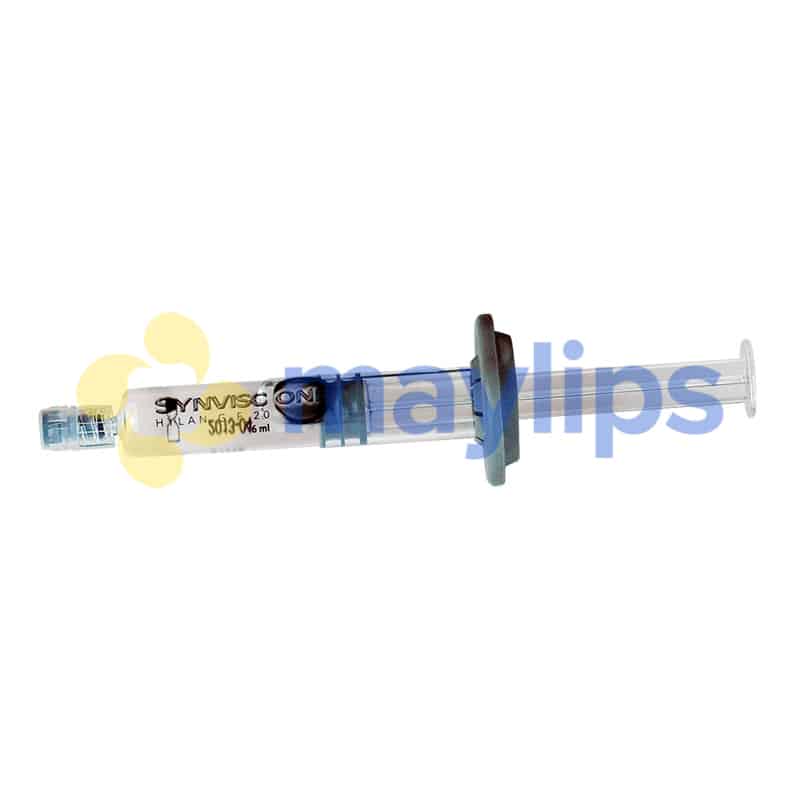
Name: SYNVISC ONE® (HYLAN G-F 20)
Ingredients: Hylan (8mg/mL), sodium chloride (8.5mg/mL), disodium hydrogen phosphate (0.16mg/mL), sodium dihydrogen phosphate hydrate (0.04mg/mL).
Size: 1x6mL pre-filled syringe.
Manufacturer: Genzyme Europe B.V.
Origin: Europe.
Also Included: Package Insert only. It is recommended to use a 18-20G range needle for injection (obtained separately).
Storage: Store between 2℃ and 30℃. Do not freeze.
Remarks: Sterile packaging.
In Stock
More info
WHAT IS SYNVISC ONE (HYLAN G-F 20)?
Synvisc One, which is sometimes also referred to as Hylan G-F 20, is an injectable orthopaedic gel made of hylan that is derived from rooster combs, the fleshy growth on top of a rooster’s head, making it a product of animal-origin. Once extracted, the hylan polymers are chemically cross-linked to make a thick, viscous, elastic, bioresorbable, and biodegradable gel that mimics the properties of naturally-occurring synovial fluid. These orthopedic injectables are specially designed to treat osteoarthritis in knee joints. The active ingredients in this implant are 8mg/mL of hylan, 8.5mg/mL of sodium chloride, 0.16 mg/ml of disodium hydrogen phosphate, 0.04mg/mL of sodium dihydrogen phosphate hydrate, and water for injection.
Synvisc One is different from Synvisc, another item in this product range, due to its packaging size, as this product is packaged in a single prefilled 6mL syringe. This single-use treatment syringe is sold together with traceability labels and a product pamphlet. Needles are not provided and must be obtained separately. Do not freeze this sterilised item, and keep it stored away from light. Ensure that it is stored in a dry, clean and cool space, in a temperature ranging from 2º C to 30º C. This orthopaedic treatment gel is manufactured by Genzyme.
WHAT IS HYLAN G-F 20 USED FOR?
Synvisc One has been formulated to treat pain and damage caused by osteoarthritis, particularly in the knee joints. Knee joints that have been affected by osteoarthritis (OA) have a diminished amount of synovial fluid in the joint spaces, and supplementing the fluid with this intra-articular implant introduces the much-needed lubricant between the bones. Your patients can then move much more freely and without crippling pain. Individuals who have not been able to curb their symptoms with oral pain medication (e.g. acetaminophen), supplements (e.g. glucosamine), mobility support gears (e.g. knee guards), or other forms of non-pharmacological treatments can opt for Synvisc One to decrease discomfort and prevent further degeneration of the knee joints.
HOW DOES THIS INJECTABLE ORTHOPAEDIC GEL WORK?
A synovial joint is characterized by its flexibility and the presence of synovial fluid. The connecting bones in this type of joint are covered by a layer of thin white articular cartilage, which secretes lubricating synovial fluid continuously into the synovial cavity. One of the main components of the fluid is hyaluronic acid. When in optimal health, patients can move the joints freely without experiencing pain or stiffness, and harmful shocks are absorbed by the cushiony fluid. Wear and tear can exhaust the cartilage, causing it to wear off. When this occurs, less fluid is produced and the bones rub against each other, causing pain and inflammation. Sometimes, some bone fragments can chip off and get deposited in the synovial space, causing even more pain and swelling. This is how chronic, degenerative osteoarthritis conditions develop.
Synvisc One was formulated as a single-dose treatment to deliver fast relief to patients. The hydrated hylan A polymers are combined with high molecular weight hylan B polymers to create a highly elastic and viscous gel. The implant is biologically formulated to be similar to synovial fluid, and provides physical support and lubricating action to affected joints. Once injected, the bones encased within the damaged joints do not grind against each other, and pain is lessened. Consequently, patients are able to perform physical activity and normal movements confidently without uncomfortable pain. Corrective procedures like knee replacement surgeries can actually be postponed thanks to this orthopaedic implant.
HOW TO INJECT SYNVISC ONE?
Evaluate your patients’ suitability for receiving intra-articular injections by conducting an examination of the damaged joint, and by obtaining their medical history. Just before injections, sanitise the proposed area of treatment and remove any fluid accumulation if necessary. Ensure that the package is unopened and still in sterile condition before use, and ensure that the syringe has been kept at the appropriate room temperature. The methodical injection techniques are as follows:
- Twist the cap on the syringe and pull off gently to minimize product leakage.
- Attach and secure an appropriately-sized needle (18G to 20G needle, depending on injection area) to the prefilled syringe containing the implant gel.
- Administer the gel into the synovial cavity of damaged joints.
- Dispose of any unused gel, used syringes, and needles safely.
The entire 6mL syringe is the recommended dosage for a knee joint in a single session. Only certified physicians with experience and skill in intra-articular injection techniques can perform this procedure safely. Use aseptic techniques throughout the procedure to reduce risk of infection. Do not inject this solution into blood vessels or synovial tissues. Failure to do so might result in serious reactions like blood clots, severe inflammation, and stroke.
HOW LONG DOES IT LAST?
The injected hylan gel is metabolized by the body, just as hyaluronic acid is, making this a temporary solution to the treatment of osteoarthritis. A single injection of Synvisc One typically provides 6 months of relief from pain and discomfort, and the effects typically take effect a month after treatment. The duration of results will vary on an individual basis. If necessary, patients can opt for another bout of treatment about six months after the first session.
IS THIS PRODUCT SAFE?
You can confidently suggest Synvisc One as one of the best injectable treatment choices for your patients, as it has been used by innumerable patients for close to 2 decades. This product has undergone important clinical trials, which attest to the efficacy and safety of this implant. However, some groups of people should steer clear of this intra-articular injection due to lack of clinical data regarding safe use, including:
- Patients who are pregnant;
- Patients who are lactating;
- Paediatric patients.
Furthermore, since this hylan gel implant is of avian origin, people with an avian allergy (e.g. egg products, poultry, feathers) are strictly contraindicated from using it. It should also not be injected into inflamed or infected skin. A complete list of precautions and contraindications can be obtained from the product pamphlet.
COMMON SIDE EFFECTS:
- Heat sensation at treated site
- Swelling at treated site
- Pain at treated site
- Redness at treated site
- Effusion or fluid accumulation at treated site
Assure your patients that such reactions usually last only for a few days, and can be improved by applying cold compresses. You can prescribe or suggest oral analgesics for better pain management until the side effects have resolved. Patients must also rest the treated joint by avoiding strenuous or high-impact physical activities, like jogging and tennis, for at least 2 days after treatment session. Patients should see a doctor immediately if they display signs of hypersensitivity/allergic reactions.
SERIOUS SIDE EFFECTS:
- Rash outbreak
- Swollen mouth and face
- Laboured breathing
- Speech impediment
- Fever
- Muscle pain
- Headaches
- Dizziness
- Nausea, which may accompanied by vomiting
- Tachycardia
- Decreased platelet count
A complete list of possible reactions can be obtained from the product pamphlet.
WHAT IS IT MADE OF?
Synvisc One is formulated using hydrated hylan A and B polymers. These hylan polymers are of animal origin, as they are derived from rooster combs, and are chemically cross-linked for amplified viscosity and elasticity. The resulting injectable gel is viscoelastic and has similar properties to the naturally-occurring synovial fluid found in our bodies. Once injected, it can provide lubrication and mechanical support to damaged joints.
Important Information
Warning! The product is intended solely for professional use in a medical environment and under professional supervision. The product is supplied to medical professionals who specialize in aesthetic medicine (plastic surgery, facial treatments) and work under individual medical licenses as aestheticians or in dedicated clinics. Unprofessional use by individuals without a medical background may lead to severe adverse effects and health harm.
















Reviews
There are no reviews yet.