You must be logged in to post a review.
ETERMIS 4
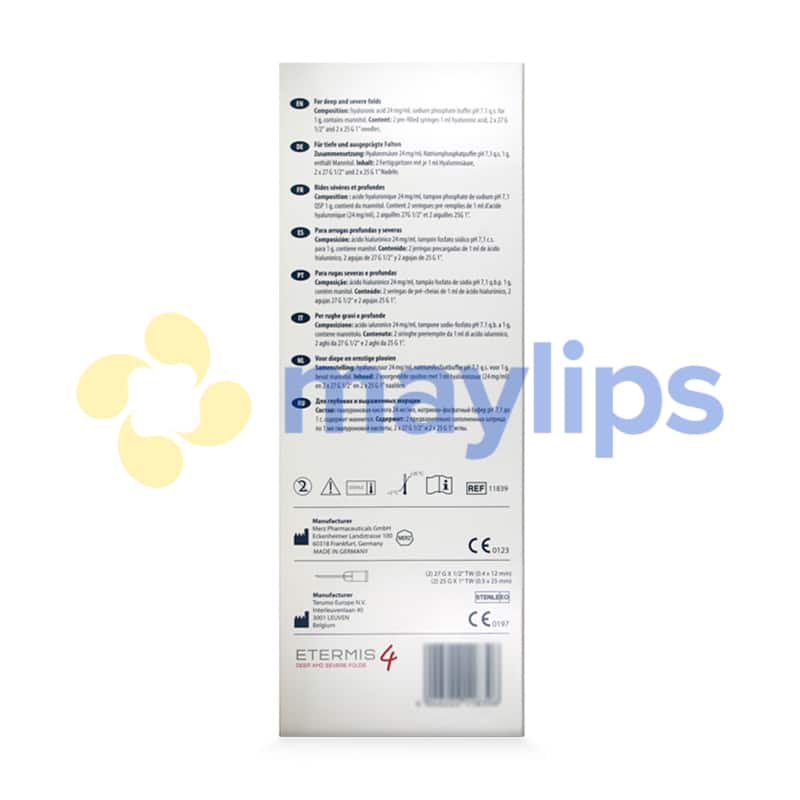
Active Compositions: 24 mg/ml hyaluronic acid, sodium phosphate buffer pH 7.1 q.s. for 1 g, mannitol.
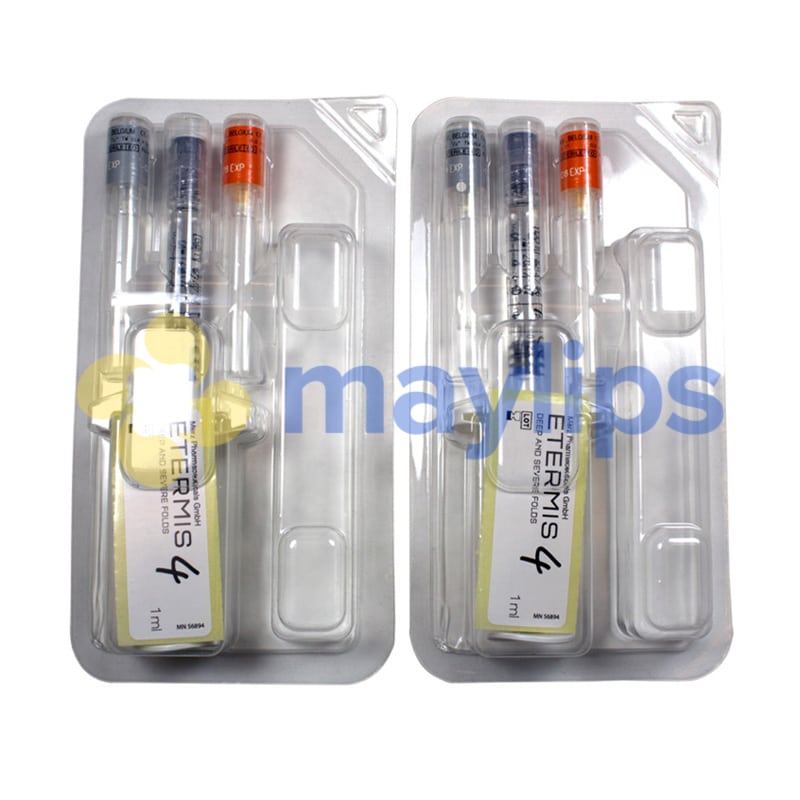
Volume & Packaging: 2 x 1ml pre-filled syringe.
Manufacturer: Merz Aesthetics.
Also Included: Contains two 27G 1/2″ needles and 2 x 25G 1″ needles, packaging insert.
Remarks: Steryl packaging.
In Stock
More info
WHAT IS ETERMIS 4?
Etermis 4 is a versatile dermal filler manufactured by Merz Aesthetics that is designed to perform lip augmentation and correct winkles. The cosmetic filler consists of 24mg/mL of hyaluronic acid combined with antioxidant mannitol contained in a sodium phosphate buffer of pH 7.1. While the hyaluronic acid smooths out lines and folds, mannitol protects the skin structure and the injected filler from further damage. The monophasic, cohesive gel is packaged with 2-1mL graduated syringes and comes together with 2 sterile 27G½” needles, 2 sterile 25G1” needles, a product leaflet and traceability labels. Keep this fragile item in a cool, dry room with a temperature between 2°C and 25°C. It should also be protected from frost, heat and direct light.
WHAT IS IT USED FOR?
Etermis 4 is designed for deep-set folds, lip augmentation, and for increasing or refining facial volume and contours.
HOW DOES THIS DERMAL FILLER WORK?
Skin cells that provide structure, hydration and nourishment (e.g. collagen, hyaluronic acid and elastin) dwindle in their quantity and quality as the body ages. Combine this natural process with external damaging factors like repetitive facial movements, prolonged sun exposure, and smoking, and you get severe wrinkles. Often times, these issues make a person look tired and older than their age.
Etermis 4 injections introduce a flexible and hydrating hyaluronic acid gel into the weak skin structure underneath wrinkles. The cohesive gel is malleable and provides a good framework for the skin. Wrinkles are filled in and skin becomes smoother. Mannitol has been added to provide a protective antioxidant property to the gel and the skin. The antioxidant stops any damaging free radicals from causing harm to the gel. As a result, the aesthetic gel can remain on the treated area for a longer time before being degraded. Post-injection inflammatory reactions are also curbed and patients’ recovery times are much shorter. Lines are filled in and patients can enjoy a radiant-looking face in next to no time.
HOW TO INJECT IT?
Etermis 4 injections are considered to be a medical procedure and can only be performed after careful reviews have been completed by certified physicians who are experienced in dermal-filler injection techniques. Evaluating patients’ medical information (e.g. allergies, medications and supplements, persisting health problems) ensures a better treatment outcome.
Make sure the cosmetic filler is at room temperature. Also, ensure that the original packaging is still intact and the item is within its shelf life. Remove the syringe aseptically from the tray. Remove the protective cap from the syringe and attach a needle (27G½” or 25G1”) to it securely.
Disinfect the proposed treatment area. You may also apply some numbing cream to reduce the patient’s pain during procedure. Inject the filler using the fanning, linear threading, serial puncture, sandwich, or cross-hatching technique. The quantity of gel depends on the severity of the skin problem and the area of treatment. Do not overcorrect.
Lightly massage the injected area for good product distribution. Dispose any unused filler gel and used needles.
All the steps above must be done aseptically in a clean environment. This filler should not be injected intravascularly or into unintended areas (e.g. nose, glabellar region, breasts, tendons, muscles, bones). Read the product leaflet for a complete list of precautions and warnings.
HOW LONG DOES THIS FACE FILLER LAST?
A single Etermis 4 injection can last for approximately a year before it disintegrates into the surrounding tissues. Its duration varies on an individual basis. Factors like skin conditions, the patient’s health status, the amount of gel used and the injecting skills of the doctors are all paramount in determining the lasting ability of the filler.
IS THIS PRODUCT SAFE?
Etermis 4 is a very safe dermal filler gel manufactured by superior Germany engineering. The hyaluronic acid is derived from a non-animal source and purified to be free from disease-causing endotoxins and pyrogens. In addition, hyaluronic acid exists naturally in various parts of the body and the chances for rejection reactions are minimal. But, it is still not recommended for the following people due to unestablished safety profile:
- Patients on certain medications such as anticoagulants, non-steroidal anti-inflammatory drugs (NSAIDs) and immune-altering medicines;
- Patients suffering from allergic reactions and autoimmune disorders;
- Patients suffering from skin disorders such as inflammations (e.g. rash, acne), infections (e.g. herpes), and hypertrophic scarring tendency;
- Patients with hypersensitivity to hyaluronic acid-based products;
- Pregnant and breastfeeding patient;
Patients whose skin is still affected from previous dermal procedures (e.g. dermabrasion, laser treatments, chemical peels) should wait until complete recovery before obtaining Etermis 4 injections. Read the product leaflet for a complete list of contraindications.
ETERMIS 4 SIDE EFFECTS
After the treatment session, your patients might notice some side effects that are usually associated with gel implantation.
COMMON SIDE EFFECTS
- Inflammatory reactions (e.g. pain, redness, swelling, itching)
- Burning or tingling sensation
- Bleeding, bruising, and blood clots
- Skin discolouration (e.g. Tyndall effect if it is injected too close to the skin surface)
- Filler gel displacement
Patients should not wear makeup for at least 12 hours following the procedure. Extended sun exposure and extreme temperatures (e.g. sauna, Turkish baths, freezing temperatures) must also be avoided for a minimum of 2 weeks after treatment.
ADVERSE REACTIONS
- Allergic reactions
- Infections
- Pus formation
- Tissue death
Advise patients that these reactions can happen immediately after procedure or be delayed. Nevertheless, patients should get medical attention if the reactions persist for more than a week. Read the product leaflet for the complete list of possible side effects.
WHAT IS IT MADE OF?
Etermis 4 is a hyaluronic acid cosmetic gel derived from non-animal sources using advanced German engineering. The gel is processed to be monophasic and cohesive. Mannitol has been integrated as well. The resulting gel is reconstituted into buffer a solution of pH 7.1.
Important Information
Warning! The product is intended solely for professional use in a medical environment and under professional supervision. The product is supplied to medical professionals who specialize in aesthetic medicine (plastic surgery, facial treatments) and work under individual medical licenses as aestheticians or in dedicated clinics. Unprofessional use by individuals without a medical background may lead to severe adverse effects and health harm.












Reviews
There are no reviews yet.