You must be logged in to post a review.
HYMOVIS® GERMAN
Product: HYMOVIS® GERMAN
Manufacturer: Fidia Pharmaceuticals
Active Substance(s): Hyaluronan
Strength: 24mg/3ml
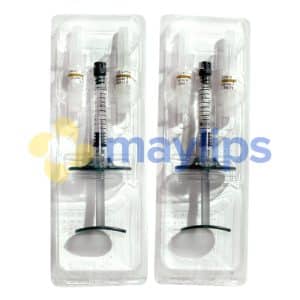
Pack Size: 2-3ml prefilled syringes
Accessories: Package insert
In Stock
More info
WHAT IS HYMOVIS?
Hymovis is an intra-articular implant that is manufactured by Fidia Pharma, a pharmaceutical organization that specializes in joint healthcare, aesthetic dermatology, and advanced wound care. This sterile, pyrogen-free, and viscoelastic implant is made of 8mg/ml of high molecular weight and ultra-pure hyaluronan that is highly modified, dissolved in physiologic saline, and is free from any chemical cross-linking. The hyaluronan is derived from bacterial fermentation and is processed via patented technology to mimic the action of naturally occurring synovial fluid to alleviate pain associated with osteoarthritis (OA). Each box of Hymovis is packaged with a package insert and 2 sterile and single-use 5mL syringes that are prefilled with hyaluronan gel. Store Hymovis in its original packaging at room temperature (25°C) away from freezing conditions.
WHAT IS IT USED FOR?
Hymovis is a nonanimal-based hyaluronan intra-articular implant that is designed to replace synovial fluid in knee joint(s) affected by OA, resulting in successful alleviation of pain associated with OA. It is best used in osteoarthritic patients who failed to manage their OA condition via lifestyle modification (e.g. weight loss, exercise therapy, rehabilitation gear, etc.) or pharmacological therapy (e.g. acetaminophen, etc.). Hymovis should be administered via a two-injection regimen into the affected knee joint. These injections should be spaced one week apart for optimal improvement of the OA.
HOW DOES IT WORK?
Synovial joints, such as the knees and hips, are characterized by the flexible synovial membrane. The ends of bones within this membrane are sheathed with a thin layer of protective articular cartilage, a collection of tough and specialized connective tissues that allow the bones to glide against each other smoothly with minimal friction. Synovial membrane is also responsible for producing lubricating synovial fluid that acts as a shock absorber in case of sudden impacts. Unfortunately, when the articular cartilage degenerates due to wear-and-tear, excessive weight, or injuries, the bones are left exposed and grind painfully against each other during movements. This condition is known as osteoarthritis (OA), which is marked by debilitating symptoms such as pain, swelling, joint stiffness, and lack of mobility.
Hymovis is made of 8mg/ml of viscoelastic hyaluronan that is sourced from biofermentation. Following extraction and purification, this hyaluronan is processed using patented technology without the use of any chemical reticulating agents. The resulting hyaluronan is known as HYADD 4 and has superior lubricating and shock absorbing properties. Once injected intraarticularly into affected knee joints, Hymvois replaces the lost synovial fluid, and patients can resume their activities of daily living with reduced pain and discomfort.
HOW TO USE IT?
A complete intra-articular implantation of Hymovis occurs in the manner detailed below:
- A pre-procedural consultation session is a must, as it helps the physician to evaluate their patient’s health status and the severity of their OA condition. This consultation session is also reserved for educating the patient about the possible side effects that they might experience and the appropriate aftercare methods that they should follow for quick recovery. Once they are deemed ready, a suitable treatment date can be set. Note that only patients without any contraindicating conditions are allowed to receive Hymovis injections.
- On the day of procedure, start the session by cleaning and sanitizing the injection area. Any joint effusion must be removed using an 18- to 20-gauge needle; failure to do so will only result in undesirable side effects. Next, prepare the medical device by securing an 18- to 20-gauge sterile needle to the tip of the prefilled syringe. Inject the full 3ml hyaluronan gel into the intra-articular space without overfilling the joint. If a patient requires treatment in both joints, then use a separate syringe for each knee joint. A week later, administer the second injection into the affected knee(s).
- Once you have administered the hyaluronan gel, discard all syringes and needles into biological waste bins.
The entirety of the procedure detailed above can only be conducted by trained and experienced medical practitioners under strict aseptic settings.
HOW LONG DOES IT LAST?
Patients should be able to notice desirable improvements in their OA condition for about six months after a treatment session with Hymovis. The duration of action of Hymovis is influenced by factors like the severity of the OA. Once this injected viscosupplement wears off and symptoms of OA start reappearing, patients can always get another Hymovis injection.
IS IT SAFE?
Yes, Hymovis is a very safe viscosupplementation, as it is made in a strict laboratory setting. Its active ingredient, hyaluronan, is derived from a tightly regulated bacterial fermentation process and then undergoes an extensive purification process. The absence of any pyrogens, chemical reticulating agents, and animal proteins make Hymovis an exceptionally safe and effective intra-articular implant. That being said, its usage is contraindicated in the following patients due to a low safety profile:
- Patients who are allergic to hyaluronan;
- Patients who are allergic to gram positive bacterial proteins;
- Patients who present dermal reactions (e.g. inflammations, injections, etc.) in the proposed area or joint of treatment.
Refer to the package insert for more details on contraindications, warnings, and precautions.
WHAT ARE THE SIDE EFFECTS?
Common side effects that can occur because of the use of Hymovis include the ones listed below:
- Redness;
- Pain;
- Swelling;
- Tingling or itching sensation;
- Joint stiffness.
Tell your patients to avoid high-impact physical activities, such as jogging and tennis, and prolonged weight bearing activities, such as standing, for at least 48 hours after they receive treatment with Hymovis to reduce the risk of negative side effects. On the other hand, patients are still at risk of developing severe reactions.
Adverse side effects that can occur because of the use of Hymovis include the ones listed below:
- Arthropathy;
- Phlebitis;
- Tendonitis;
- Paraesthesia;
- Pruritus.
The lists above are not exhaustive; refer to the package insert for a detailed list of side effects.
WHAT IS IT MADE OF?
Hymovis is a hyaluronan intra-articular implant that is designed to be injected into the synovial space to alleviate pain associated with OA. Each 5mL syringe is preloaded with sterile gel that contains 8mg/ml of high molecular weight, ultra-pure hyaluronan that is free from any chemical cross-linking; this hyaluronan is dissolved in physiologic saline. Hymovis should be injected in a two-injection regimen in an affected knee joint; each injection is meant to be administered one week apart for optimal results.
Important Information
Warning! The product is intended solely for professional use in a medical environment and under professional supervision. The product is supplied to medical professionals who specialize in aesthetic medicine (plastic surgery, facial treatments) and work under individual medical licenses as aestheticians or in dedicated clinics. Unprofessional use by individuals without a medical background may lead to severe adverse effects and health harm.













Reviews
There are no reviews yet.