You must be logged in to post a review.
TEOSYAL® RHA4
Name: TEOSYAL® RHA 4.
Ingredients: Cross-linked hyaluronic acid (23mg), Lidocaine hydrochloride (3mg), Phosphate buffer pH 7.3 (qs ad 1ml).
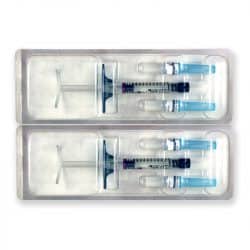
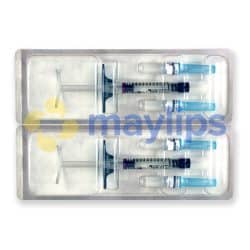
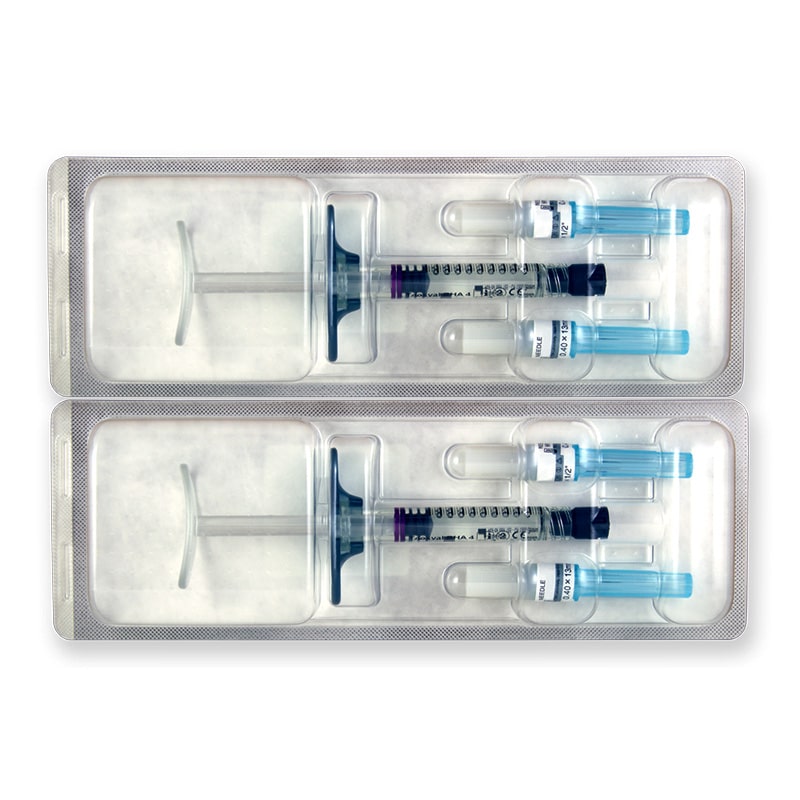
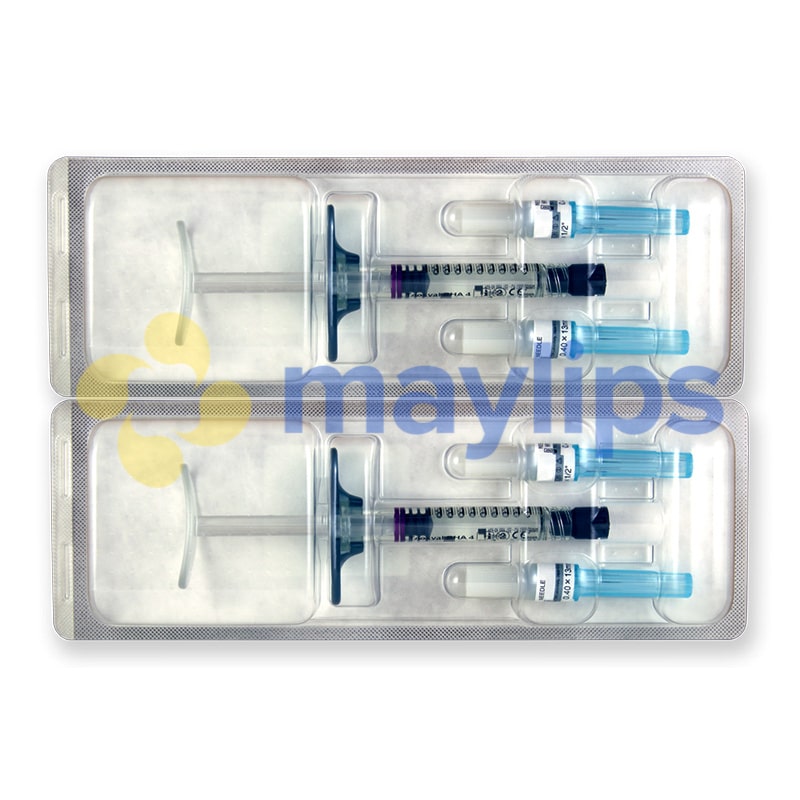
Size: 2 x 1ml pre-filled syringes.
Manufacturer: Teoxane Laboratories.
Origin: Europe.
Also Included: For each syringe, the box contains two sterile 27G 1/2″ needles, two traceability labels, packaging insert.
Storage: Store between 2℃ and 25℃.
Remarks: Sterile packaging.
In Stock
More info
WHAT IS TEOSYAL RHA 4?
Teosyal RHA 4 is a sterile, transparent, non-pyrogenic, viscoelastic gel of cross-linked, non-animal hyaluronic acid. It also contains 0.3% lidocaine hydrochloride by mass for anaesthetic properties.
This dermal filler is part of Teoxane Laboratories’ Teosyal RHA line, which includes four separate formulas for different patient needs. This innovative range was created according to a patented process, making it possible to meet the specific requirements of mobile facial areas.
A study conducted between Teoxane Laboratories and the University of Grenoble found that under both stretching and compression, this dermal filler adapted to its environment and maintained its performance. With its unparalleled stretch and strength properties, this dermal filler was also found to improve wrinkles based on the Wrinkle Severity Rating Scale, improve overall aesthetics on the Global Aesthetic Improvement Scale, and to cause great patient satisfaction at 88.6%.
Each box of Teosyal RHA 4 contains two prefilled syringes. It also holds two sterile 27G½” needles and two traceability labels, one which should be given to the patient, and one to be kept by the doctor. The volume of each syringe is shown on the box as well as on each syringe.
In general, one or two treatment sessions are necessary, depending upon the depth of the wrinkles and/or volume in need of treatment. Periodic touch-up sessions enable the patient to upkeep their sought-after results.
It should be stored between 2°C and 25°C, away from direct sunlight.
WHAT IS IT USED FOR?
Teosyal RHA 4 is used to add volume to the face, helping to modify the anatomy of aged skin, fill depressions and wrinkles, and restore skin’s natural hydration levels. Specifically, it is used for augmenting the cheeks and cheekbones, redefining facial contours, correcting the lower eyelids and minor ptosis, and filling deep wrinkles in thick skin on the face.
This cosmetic filler may also be used for the purposes of reconstructive surgery. This includes the restoration of volume lost due to lipoatrophy and the filling of depressions caused by scarring.
HOW DOES THIS DERMAL FILLER WORK?
Teosyal RHA 4 features an innovative, resilient form of hyaluronic acid. Known as the “Preserved Network” method, Teoxane Laboratories has created a new form of this natural chemical that features specific cross-linking conditions that better preserve the length of hyaluronic acid chains. This helps to maintain the skin’s natural architecture, sustaining desired volume and structure.
HOW TO INJECT
One week before the injection session, the patient should be advised not to take high-dose vitamin E, aspirin, anti-inflammatory drugs or anti-coagulants.
To lessen the occurrence of potential complications, Teosyal RHA 4 should only be used by an authorized medical practitioner trained in subcutaneous and/or preperiosteal injection techniques for adding volume to the subcutaneous facial tissue. This dermal filler should be injected using 27G½” hypodermic needles, like those supplied in the box.
Before starting the injection, the treated area must be thoroughly cleaned and disinfected with a suitable antiseptic solution. Any makeup products should be removed, and natural oils should be wiped away.
Inject the product slowly, taking care to advance the needle along the desired trajectory. Injection techniques may vary according to both the quantity of solution administered, as well as the treatment area. Please note that while the injection volume is dependent upon the required level of correction, the amount should not exceed 4mL per session. To prevent the filler from flowing out of the treatment site, stop the injection well before withdrawing the needle from the skin. Avoid injecting too superficially, as this may cause irregular and unwanted results.
After the injection, massage the treated sites carefully in order to ensure a uniform distribution of Teosyal RHA 4. If slight swelling occurs, apply melted ice to the site for a few moments. It is important not to overcorrect the affected area. The patient should not use makeup for up to 12 hours following the injection, and should avoid extreme temperatures for one week following treatment.
At the end of the treatment session, it is imperative to discard all remaining unused product. Moreover, this device is intended for single use only; sterility is not guaranteed in the event of re-use. The gel can deteriorate or dry out, reducing its efficiency and hindering its extrusion through a needle. Dispose of used needles in appropriate containers.
HOW LONG DOES IT LAST?
While results may vary from patient to patient, Teosyal RHA 4 has been found to last between 6 and 18 months.
IS THIS PRODUCT SAFE?
Teosyal RHA 4 has been extensively tested to ensure its safety. However, patients with known allergies to hyaluronic acid, lidocaine, or amide-type anaesthetic, as well as those with poryphria, hepatocellular insufficiency, liver disease, or epilepsy should not use Teosyal RHA 4. Additionally, those who are pregnant, breastfeeding, or under the age of 18 should not be injected with dermal fillers.
It should be noted that due to possible interactions with other filling implants, this face filler should not be introduced in to sites that have been previously injected with cosmetic fillers. Athletes must also be aware that Teosyal RHA 4 contains an active ingredient that may cause a positive result in a drug test. Moreover, this cosmetic filler should not be injected directly into the bloodstream.
TEOSYAL RHA 4 SIDE EFFECTS
The use of Teosyal RHA 4 may result in minor injection-related reactions, including inflammatory reactions and pain. However, these side effects generally resolve themselves within one week post-injection.
OTHER COMMON SIDE EFFECTS INCLUDE:
- Oedema
- Erythema
- Itching
- Temporary loss of sensitivity
POSSIBLE ADVERSE EFFECTS INCLUDE:
- Dyschromia
- Abscesses
- Skin necrosis
- Allergy
WHAT IS IS MADE OF?
Teosyal RHA 4 features a hyaluronic acid base, which is naturally produced in the body. Hyaluronic acid has an innate ability to bind and retain water molecules, making it a necessary component in the maintenance of hydrated, bright, and healthy skin.
Unfortunately, as the skin ages, its hyaluronic acid content depletes. This causes loss of elasticity, smoothness, and volume in the face, creating sallow, wrinkled skin. By injecting hyaluronic acid back into the skin—such as that found in Teosyal RHA 4—it appears more plump, hydrated, and radiant.
This facial filler also contains lidocaine, which is meant to improve overall patient comfort. This local anaesthetic is used to treat any minor discomfort associated with cosmetic procedures, working to decrease pain by temporarily numbing the injection site.
Important Information
Warning! The product is intended solely for professional use in a medical environment and under professional supervision. The product is supplied to medical professionals who specialize in aesthetic medicine (plastic surgery, facial treatments) and work under individual medical licenses as aestheticians or in dedicated clinics. Unprofessional use by individuals without a medical background may lead to severe adverse effects and health harm.


















Reviews
There are no reviews yet.