You must be logged in to post a review.
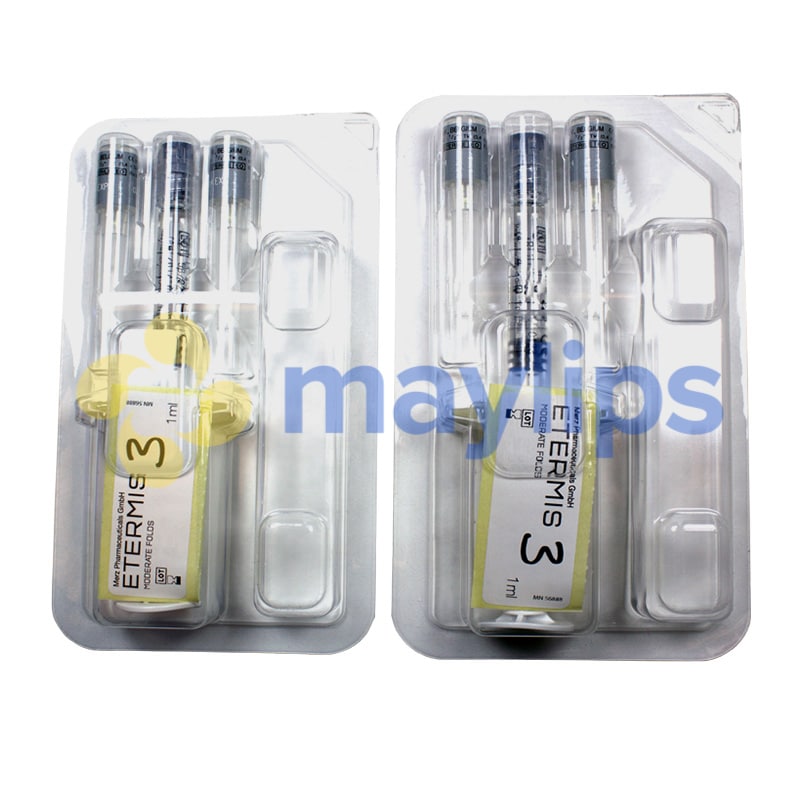
ETERMIS 3
Active Compositions: 23 mg/ml hyaluronic acid, sodium phosphate buffer pH 7.1 q.s. for 1 g, mannitol.
Volume & Packaging: 2 x 1ml pre-filled syringe.
Manufacturer: Merz Aesthetics.
Also Included: Contains four 27G 1/2″ needles, packaging insert.
Remarks: Steryl packaging.
In Stock
More info
WHAT IS ETERMIS 3?
An injectable gel made from hyaluronic acid, mannitol, and lidocaine, Etermis 3 offers patients an effective method for reducing fine lines, wrinkles, and thinning of the lips. Most generally, this face filler is suitable for use on skin with early signs of aging.
Part of Merz Aesthetics’ line of Etermis fillers, Etermis 3 has excellent patient tolerability and a low rate of adverse events. Complete with 3 different forms of injectable filler, this range offers a variety of aesthetic applications, including the enhancement of the lips, the reversal of deep folds, and the reduction of fine lines and wrinkles.
Merz Aesthetics is a leader in the aesthetic and therapeutic markets of both Europe and North America. The creator of the world’s first anti-wrinkle cream in 1953, this company is a driver of innovation and development in the field of aesthetic medicine. Patients and practitioners can rest assured knowing that Merz products are backed by decades of research, development, and distribution of cosmetic products
Etermis 3 comes in sterile packaging, and includes 2 prefilled 1mL syringes and 4 sterile 27G ½” needles. This gel should be stored between 2 and 25°C, and should be kept away from light and frost to avoid degradation.
WHAT IS IT USED FOR?
Etermis 3 is used to reduce moderate wrinkles and offer enhancement to the lips. Specifically, it can be injected into the nasolabial folds and marionette lines in order to give the face a more youthful appearance.
HOW DOES THIS DERMAL FILLER WORK?
Made with sodium hyaluronate—also known as hyaluronic acid— Etermis 3 has incredible hydrating and volumizing abilities when injected into the skin. This is because hyaluronic acid has the ability to bind with up to 1,000 times its weight in water, instantly adding cushion to the skin cells and the spaces between them. This adds support and volume to the tissue, helping to create a more smooth, flawless appearance. Also, because Etermis 3 is formulated in a monophasic manner, it is more cohesive than other hyaluronic acid-based fillers, meaning that it will not migrate substantially following the injection process.
This facial filler also contains mannitol, an antioxidant that works to minimize the degradation of hyaluronic acid. With this, patients are able to enjoy their desired effects for longer periods of time. Additionally, this dermal filler has a low extrusion force, making its gel easier for practitioners to inject.
HOW TO INJECT?
Etermis 3 is to be administered only by registered and authorized medical practitioners. This aesthetic filler should only be injected into non-inflamed, properly disinfected, healthy skin.
Before beginning the injection process, remove any makeup or oil from the patient’s face with soap and water, and sterilize the area with antiseptic solution. Then, gently press the plunger rod until gel is visible at the mouth of the needle.
Inject the needle into the mid to deep dermis, keeping the needle at an angle of approximately 30–35°. Push the filler slowly and gently into the affected area, being careful to avoid overcorrection and intravascular injection. To properly disperse the gel into the surrounding tissue, massage the injection site gently and thoroughly. If superficial injection occurs and causes blanching, rub the affected area until colour returns to the skin.
After undergoing treatment with Etermis 3, the patient must avoid exposure to sun and extreme cold, as ultraviolet radiation, heat, and freezing temperatures may cause the gel to degrade. Additionally, he or she should avoid strenuous exercise for 24 hours after treatment, as this can cause additional bruising. If needed, the patient may apply a cold compress to the affected area or take acetaminophen for pain, bruising, or swelling.
Remember to properly dispose of any remaining gel and used needles at the completion of the procedure. This gel may not be resterilized or reused.
HOW LONG DOES IT LAST?
The longevity of Etermis 3’s results may vary from patient to patient. This may depend on the injection site, the amount of filler used, and the patient’s age. Typically, the results can last up to a year.
IS THIS PRODUCT SAFE?
While Etermis 3 has been approved for safe use, certain patients may not use this dermal filler. Pregnant or breastfeeding women and those under the age of 18 cannot be injected with this cosmetic filler. Additionally, those with any sign of infection, clinical inflammation, or allergies to hyaluronic acid or mannitol should not undergo treatment.
ETERMIS 3 SIDE EFFECTS
While side effects may occur post-injection, they are usually very minor and often resolve themselves within 7 days. Any issues lasting longer than a week should be brought to the attention of a physician.
COMMON SIDE EFFECTS INCLUDE
- Minor discomfort or bruising
- Itching
- Swelling
- Sensitivity
POSSIBLE ADVERSE EFFECTS INCLUDE
- Allergy
- Discolouration of the injection site
- Hypersensitivity
- Erythema
WHAT IS IT MADE OF?
Etermis 3 is made with hyaluronic acid, a lubricating fluid that exists naturally in the tissues, skin, and joints of the body. Here, it helps the skin to remain hydrated, maintain collagen levels, and stay elastic. Due to these incredible effects, hyaluronic acid has become a point of obsession in the cosmetics industry, popping up in skin creams, supplements, and serums. However, this compound is most effective at maintaining skin’s youth and health in injectable fillers like Etermis 3.
This dermal filler also contains mannitol, an antioxidant used to maintain the gel’s structural integrity, decrease redness, and reduce any post-injection swelling. Naturally, mannitol is found in a variety of fruits and vegetables, as it is commonly produced by the hydrogenation of fructose. Mannitol has recently become a star player in the medical world due to its antioxidant properties. However, patients with congestive heart failure, active cerebral haemorrhage, and anuria should not be treated with mannitol.
Important Information
Warning! The product is intended solely for professional use in a medical environment and under professional supervision. The product is supplied to medical professionals who specialize in aesthetic medicine (plastic surgery, facial treatments) and work under individual medical licenses as aestheticians or in dedicated clinics. Unprofessional use by individuals without a medical background may lead to severe adverse effects and health harm.














Reviews
There are no reviews yet.